Motus Pro: Empowering Results
Motus Pro is engineered for performance, delivering next-level power with spot sizes up to 24mm and pulse durations as short as 2ms. This combination allows for exceptionally fast treatments, enabling clinics to serve more clients in less time.
From addressing pigmentation concerns and vascular lesions to performing skin tightening and rejuvenation procedures, this dual-wavelength laser provides comprehensive skincare solutions, delivering results that meet the highest standards.
Backed by science and trusted by professionals, the Motus Pro has secured FDA approval and earned its place on the NHS Supply Chain, highlighting both its safety and performance.
Its dual modes – traditional and pain-free Moveo – offer unparalleled flexibility, making it suitable for a broad spectrum of treatments across all skin tones.
Esteemed medical institutions, like Alder Hey Children’s Hospital, have praised the Motus Pro’s technology for its virtually pain-free experience, especially in treating sensitive patients. This endorsement, coupled with its advanced technological capabilities, positions the Motus Pro as the premier choice for clinics offering cutting-edge, client-centric care.
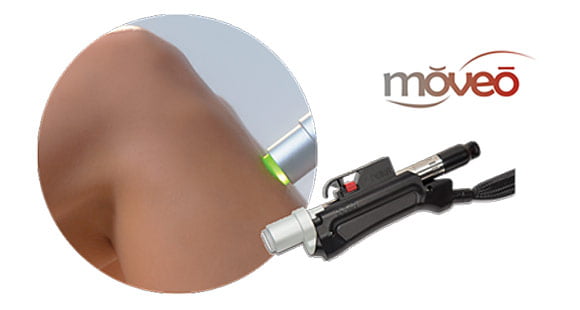
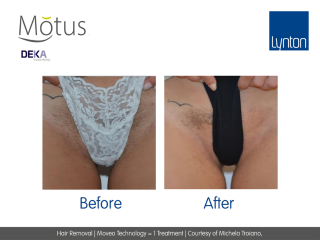
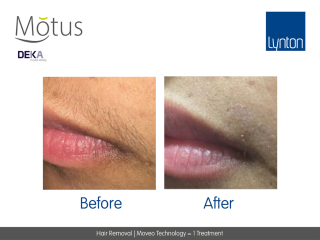
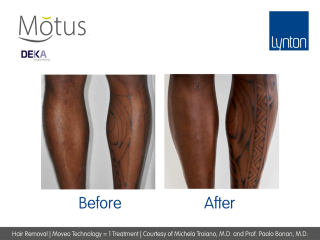
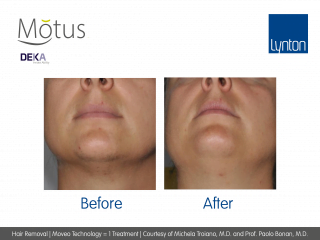
Moveo Technology

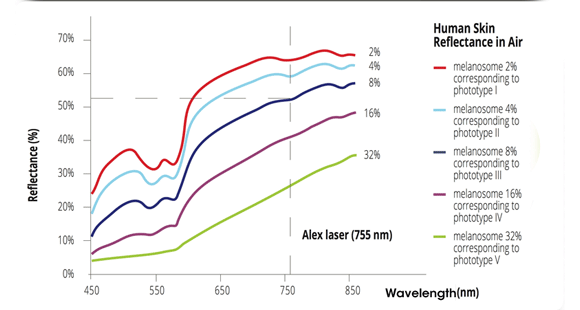
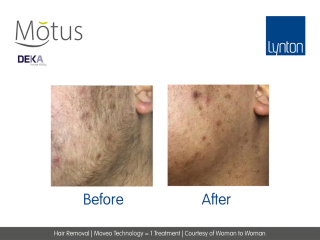
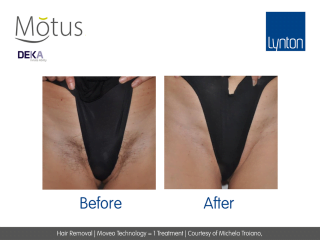
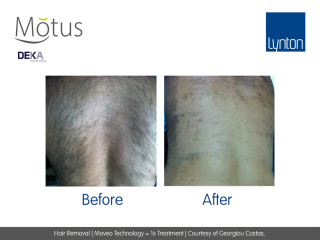
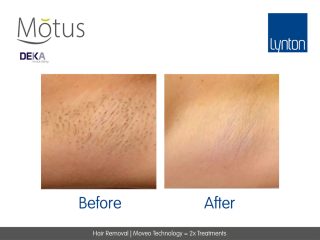
Moveo hair removal technology allows you to effectively treat skin tones 1-6 year-round using Alexandrite laser, the gold standard for hair removal.
It operates by making repeated passes over the treated areas and gradually heating the hair follicles. This multi-pass action allows for quick treatment of large areas.
The integrated sapphire cooling tip minimises risks to the epidermis while maintaining heat in the dermis where the hair follicles are targeted, ensuring a virtually pain-free and comfortable experience without compromising on excellent clinical results.
Introducing MoveoGlo: Treat Pigmented Lesions, Vascular Concerns and More With Moveo Technology

In partnership with DEKA, Lynton has introduced a new range of Moveo handpieces to the Motus Pro that extends the advantages of Moveo technology to the treatment of a wider variety of concerns, including benign Pigmented Lesions, Vascular Lesions and Skin Rejuvenation in addition to the Hair Removal handpiece that is already available. Using any one or a combination of these handpieces enables you to perform the MoveoGlo combination treatment for multiple concerns in a single session.

- Moveo Pigmented Lesions (PL) uses progressive heating to target and improve pigmented lesions such as skin age spots and sun spots.

- Moveo Skin Rejuvenation (SR) utilises the multipass technique to cause gradual heating at the dermal level, encouraging neocollagenesis.

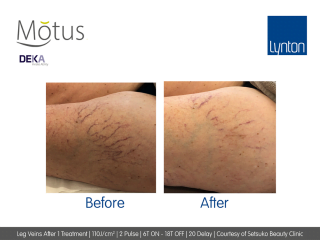
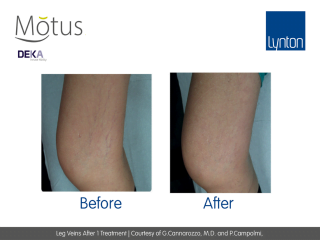
- Moveo Vascular Lesions (VL) specifically targets vascular redness such as telangiectasias for safe and effective reduction. When compared to the traditional “single spot” technique, the NEW handpieces for the treatment of pigmented lesions guarantees excellent efficacy without causing discolouration or hypopigmentation.
Other Treatment Options With Motus Pro
The Motus Pro Alexandrite and Nd:YAG Laser is a multifunctional machine that gives you the opportunity to treat a wide variety of dermatology concerns with one system. This includes: